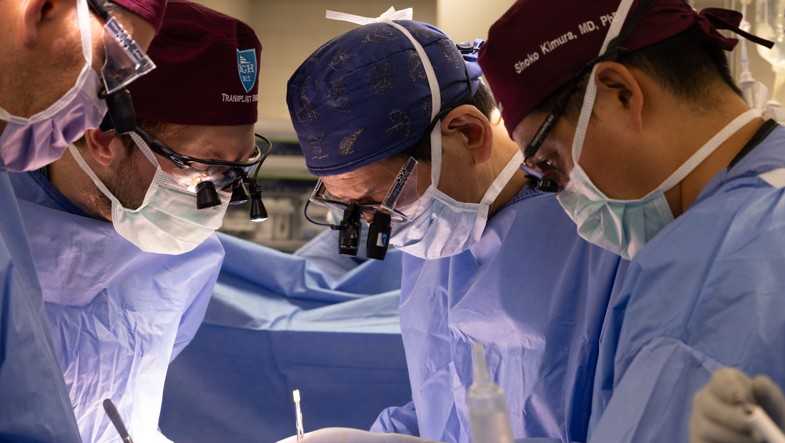
More than a year after performing the first-ever successful transplant of a genetically-engineered pig kidney into a living human patient, a Boston hospital now has the green light to take the procedure nationwide. On Monday, Mass General Brigham and Cambridge-based eGenesis announced that the Food and Drug Administration cleared the way for the start of the first clinical trial involving porcine-derived kidneys and patients with end-stage kidney disease. That approval allows the procedure to expand to transplant centers across the country and involve more than thirty patients. “It’s a big milestone,” Dr. Leonardo Riella, medical director for kidney transplantation at MGH, told NewsCenter 5 shortly before the announcement. “We suddenly have centers in different parts of the country that would be able to do these transplants. I think this would allow us really to be going the right direction so that the organ shortage doesn’t become this bottleneck that it currently is.”MGB and eGenesis also announced that the team performed their third pig kidney transplant in June. The patient, 54-year-old Bill Stewart of New Hampshire, is now back home and off dialysis for the first time in over two years. “I really wanted to contribute to the science of it,” Bill Stewart, an athletic trainer from Dover, New Hampshire, told The Associated Press.The first patient to receive a genetically-modified porcine kidney at Mass General last year, Rick Slayman of Weymouth, died less than two months after the procedure. MGB said he suffered a “cardiac event.”The second transplant patient, 67-year-old Tim Andrews, just reached the seven-month mark since the procedure. In June, he threw out the first pitch at Fenway Park. “It’s a big development forward because suddenly we have a potential alternative to dialysis to offer our patients,” said Riella. “It’s still under a research environment, but I want to frame it to all our patients who have renal failure that hope is out there.”Riella said it could be several years before the procedure is widely available to patients with renal failure. More than 800,000 Americans suffer from end-stage kidney disease. According to eGenesis, just 28,000 kidney transplants were performed last year due to a shortage of organs.
BOSTON —
More than a year after performing the first-ever successful transplant of a genetically-engineered pig kidney into a living human patient, a Boston hospital now has the green light to take the procedure nationwide.
On Monday, Mass General Brigham and Cambridge-based eGenesis announced that the Food and Drug Administration cleared the way for the start of the first clinical trial involving porcine-derived kidneys and patients with end-stage kidney disease.
That approval allows the procedure to expand to transplant centers across the country and involve more than thirty patients.
“It’s a big milestone,” Dr. Leonardo Riella, medical director for kidney transplantation at MGH, told NewsCenter 5 shortly before the announcement. “We suddenly have centers in different parts of the country that would be able to do these transplants. I think this would allow us really to be going the right direction so that the organ shortage doesn’t become this bottleneck that it currently is.”
MGB and eGenesis also announced that the team performed their third pig kidney transplant in June. The patient, 54-year-old Bill Stewart of New Hampshire, is now back home and off dialysis for the first time in over two years.
“I really wanted to contribute to the science of it,” Bill Stewart, an athletic trainer from Dover, New Hampshire, told The Associated Press.
The first patient to receive a genetically-modified porcine kidney at Mass General last year, Rick Slayman of Weymouth, died less than two months after the procedure. MGB said he suffered a “cardiac event.”
The second transplant patient, 67-year-old Tim Andrews, just reached the seven-month mark since the procedure. In June, he threw out the first pitch at Fenway Park.
“It’s a big development forward because suddenly we have a potential alternative to dialysis to offer our patients,” said Riella. “It’s still under a research environment, but I want to frame it to all our patients who have renal failure that hope is out there.”
Riella said it could be several years before the procedure is widely available to patients with renal failure.
More than 800,000 Americans suffer from end-stage kidney disease. According to eGenesis, just 28,000 kidney transplants were performed last year due to a shortage of organs.