Table of Contents
Latin America Heart Lung Machine Market Size & Trends
The Latin America heart lung machine market size was estimated at USD 99.72 million in 2024 and is projected to reach USD 150.64 million by 2033, growing at a CAGR of 4.71% from 2025 to 2033. The industry is driven by the rising prevalence of cardiovascular diseases, the increasing number of cardiac surgeries, the growing adoption of advanced cardiopulmonary bypass technologies, and the expanding healthcare infrastructure in public and private hospitals across Latin America.
Key Market Trends & Insights
The Brazil heart lung machine market accounted for the largest share of 34.68% in Latin America in 2024.
Based on product, the consumables segment held the largest share in 2024.
Based on application, the Coronary Artery Bypass Grafting (CABG) segment held the largest share in 2024.
Based on end use, the public healthcare institutions segment held the largest share in 2024.
Market Size & Forecast
2024 Market Size: USD 99.72 Million
2033 Projected Market Size: USD 150.64 Million
CAGR (2025-2033): 4.71%
Brazil: Largest market in 2024
In February 2025, Lancet Regional Health Americas reported that in 2021, Latin America and the Caribbean accounted for a growing share of the region’s cardiovascular disease burden, with CVD causing 1.1 million deaths in the Americas and ischemic heart disease and stroke as the leading causes.
The expansion of heart and lung transplantation programs in Latin America is significantly increasing demand for advanced perfusion and extracorporeal support technologies, such as heart-lung (HL) machines and related consumables. According to a recent registry, 12,374 heart transplants were conducted at 166 centers across 16 Latin American countries between 1968 and December 2022. This procedural volume reflects a maturing transplant infrastructure in the region, which in turn is driving further need for perfusion equipment and services.
Moreover, lung transplantation is on the rise in Latin America. For instance, Brazil’s national registry reported 78 lung transplants performed in 2023 across three states (Rio Grande do Sul, Rio de Janeiro, and São Paulo), equating to 0.4 transplants per million population. This figure illustrates growing capacity and a positive trend in advanced organ transplantation-a promising sign for the heart-lung machine market, particularly for devices offering dual heart-lung support or advanced perfusion capabilities. Similarly, the Christus Muguerza hospital in Monterrey is the only active lung transplant program in Mexico, performing 35 transplants between August 2017 and March 2023.
The requirements of heart and lung transplant programs drive demand for high-specification equipment and consumables, as these procedures often involve extended perfusion times, combined organ transplants (such as heart lung), and advanced monitoring needs. Furthermore, the Latin America heart transplant registry reports combined organ transplants occurring in 62.5% of participating countries. These more complex cases result in higher equipment usage and increased consumption of disposables (such as oxygenators, perfusion circuits, and cannulae).
Heart Transplantations in Latin America by Country (as of December 2022)
Country
Total Heart Transplantations
Adult Heart Transplantations
Pediatric Heart Transplantations
Argentina
2965
2672
293
Bolivia
1
1
0
Brazil
5931
5339
592
Chile
Complete Analysis will be part of Final Deliverable
Colombia
Costa Rica
Cuba
Dominican Republic
Ecuador
El Salvador
Guatemala
Honduras
Mexico
Nicaragua
Panama
Paraguay
Uruguay
Venezuela
Total
Furthermore, collaboration between device manufacturers and hospitals is the prominent driver of growth in the market, as it reduces the time from prototype to clinical use. Major manufacturers and specialist perfusion companies, such as LivaNova PLC and Getinge, conduct targeted clinical trials and regulatory programs in collaboration with cardiac centers to validate new cardiopulmonary bypass and ECMO platforms in real-world settings. Achieving regulatory milestones and generating early clinical data directly support broader procurement and hospital adoption.
Moreover, in regulatory validation, co-development partnerships often involve joint clinical studies and the development of clinical workflows. These efforts support the integration of new hardware and monitoring software into perfusion workflows with clear outcomes. Academic and clinical trials of tailored perfusion strategies or minimized bypass circuits have demonstrated reduced inflammatory responses and fewer complications when devices and protocols are co-designed and tested in hospitals. This evidence encourages hospitals to invest in new systems and supports broader clinical acceptance.
Technological partnerships and collaborations
Company Name
Year
Month
Description
Eurosets & Fundación Clínica Shaio
2025
May
Organized “Perfusion Day LATAM” in Bogotá, Colombia a training event co-hosted by Eurosets (manufacturer/educator) and Fundación Clínica Shaio aimed at up-skilling perfusionist teams on ECMO/CPB advanced monitoring and regional best-practice.
The growth of pediatric and neonatal cardiac surgery programs in Latin America presents a significant market opportunity for the heart-lung machine market. Previously, pediatric cardiac care was limited to a few tertiary hospitals. Over the past decade, capacity-building initiatives led by NGOs and government agencies have significantly improved access to surgeries for congenital heart defects (CHDs). For example, Children’s HeartLink has established long-term partnerships in Brazil with Hospital da Criança e Maternidade (São José do Rio Preto), Hospital de Messejana (Fortaleza), and Instituto do Coração (InCor) in São Paulo. In 2023, the São José do Rio Preto center became Children’s HeartLink’s first center in Latin America, recognizing its expertise in complex pediatric cardiac surgeries and staff training. These efforts have driven consistent demand for pediatric-specific heart-lung machines, including neonatal oxygenators, small-bore circuits, and precise low-flow control features designed for infants and newborns.
The rapid expansion of ECMO therapy across Latin America during and after COVID-19 has created a strong growth catalyst for heart-lung machine manufacturers, as ECMO relies on the same core perfusion components-pumps, oxygenators, and circuits-used in cardiac bypass. Many tertiary centers in Brazil, Colombia, Chile, and Mexico scaled ECMO capacity significantly between 2020 and 2023, with ELSO reporting a multi-fold increase in regional registry participation, indicating lasting institutional investment. The rise of mobile and transport ECMO programs, including air-transfer deployments using systems like Getinge Cardiohelp, has further driven standardization of perfusion consumables and cross-compatibility between ECMO and surgical bypass workflows. As hospitals transition ECMO from crisis response to routine practice, this expanding infrastructure continues to boost long-term demand for bypass-compatible equipment, creating a high-value cross-sell opportunity for heart-lung machine vendors.
Market Concentration & Characteristics
The industry exhibits a significant degree of innovation, driven by the adoption of portable and next-generation cardiopulmonary support devices. For instance, in November 2025, the Caribbean Heart Institute in Guyana deployed a portable ECMO (extracorporeal membrane oxygenation) life-support system, enhancing treatment access for patients with severe cardiac and pulmonary failure. This development strengthens critical cardiac care capabilities in the region reflects the broader trend toward compact, efficient, and high-performance machines that improve patient outcomes and operational flexibility across hospitals and cardiac centers in Latin America.
Players in the Latin America industry engage in moderate mergers, acquisitions, and strategic procurement initiatives to expand technological capabilities and strengthen regional market presence. For instance, in June 2025, Pequeno Príncipe Hospital in Brazil acquired two S5 HLM (heart-lung) machines for cardiopulmonary bypass procedures. This acquisition, funded by the State Government, enhances surgical safety and pediatric care through advanced monitoring and blood management features, making the hospital the third in Brazil to adopt these next-generation devices.

The Latin American industry is governed by comprehensive regulatory frameworks to ensure device safety, clinical performance, and patient protection. Oversight focuses on cardiopulmonary bypass systems, oxygenators, blood management devices, and advanced perfusion monitoring equipment. Regulatory compliance affects device design, clinical testing requirements, manufacturing standards, and market approval timelines. Agencies are increasingly facilitating the adoption of emerging technologies, such as compact ECMO systems, AI-assisted perfusion monitoring, and advanced blood conservation features, which enhance surgical efficiency and patient outcomes. Strict adherence to regulatory standards remains essential for market access, device reliability, and the successful integration of next-generation heart-lung machines across hospitals and cardiac care centers in Latin America.
Key players in the Latin American industry are actively broadening their product portfolios to meet rising demand for advanced cardiopulmonary support technologies. Manufacturers are introducing innovations such as next-generation heart-lung machines with integrated blood management systems, compact and portable ECMO devices, advanced oxygenators, and AI-enabled perfusion monitoring solutions. These enhancements aim to improve patient safety, optimize surgical outcomes, enhance procedural efficiency, and expand access to high-quality cardiopulmonary care across hospitals, pediatric centers, and specialized cardiac care facilities in the region.
Key players in the Latin America industry are strategically expanding their presence across the region, including Brazil, Mexico, and other emerging markets, at a moderate pace to meet the rising demand for cardiopulmonary support and advanced surgical care. Expansion efforts focus on strengthening distribution networks, hospital partnerships, clinical training programs, and remote perfusion monitoring services to improve access, device utilization, and patient outcomes. Companies are also introducing next-generation heart-lung machines, portable ECMO systems, advanced oxygenators, and integrated blood management solutions to address diverse clinical needs and support the adoption of innovative cardiopulmonary technologies across Latin America.
Product Insights
By product, the consumables segment led with the largest revenue share of 61.10% in 2024. Segment encompasses critical single-use components required for cardiopulmonary bypass and ECMO procedures, including oxygenators, cannulas, tubing circuits, and blood reservoirs. Among these, membrane oxygenators with microporous hollow fiber technology are widely utilized; blood passes over thousands of hollow fibers, allowing efficient oxygen delivery and carbon dioxide removal while minimizing blood trauma. Modern designs prioritize high surface area-to-volume ratios and low resistance, ensuring consistent performance for both adult and pediatric surgeries. Emerging trends in this segment include miniaturized pediatric oxygenators, improved biocompatible coatings to reduce inflammatory responses, and integration of sensors for real-time monitoring of perfusion parameters.
The capital equipment segment is expected to grow at a significant CAGR over the forecast period. The segment comprises essential devices, such as pumps, heater-cooler units/heat exchange systems, monitoring systems, and other supporting equipment used during cardiopulmonary bypass and ECMO procedures. Technological advancements have led to pumps that are more compact, energy-efficient, and compatible with diverse perfusion circuits, enabling precise control, ease of operation, and adaptability during complex cardiac surgeries. Heater-cooler units and heat exchange systems regulate patient temperature, while advanced monitoring systems provide real-time physiological feedback, ensuring patient safety and optimal surgical outcomes.
End Use Insights
By end use, the public healthcare institutions segment accounted for the largest revenue share of 55.20% in 2024. Public healthcare institutions include government hospitals, regional cardiac centers, and teaching hospitals that deliver cardiac surgery and perfusion services under publicly funded programs. These institutions emphasize high-volume, cost-effective care while adhering to regulatory and quality standards. In April 2023, Hospital de Messejana Dr. Carlos Alberto Studart Gomes held an online exchange under the BRICS Visiting Scholar Program, showcasing its expertise in cardiac care, including over 500 HT, and exploring opportunities for international collaboration in cardiovascular research and training.
The private healthcare institutions segment is expected to grow at the fastest CAGR over the forecast period. Private healthcare institutions include specialty cardiac hospitals, multi-specialty private hospitals, and high-end cardiac centers performing elective and complex cardiac procedures. These institutions prioritize advanced technology, patient comfort, and reduced procedure times. In September 2023, an article in Air Ambulance Review described the successful air transport of a 57-year-old Brazilian patient under ECMO support from Campo Grande to São Paulo. The report highlighted the complexity of managing critically ill patients with lung failure over long distances in Brazil, emphasizing the importance of specialized teams, advanced equipment, and precise logistics to ensure safe outcomes.
Application Insights
By application, the coronary artery bypass grafting (CABG) segment accounted for the largest revenue share of 31.33% in 2024. CABG is a standard treatment for multivessel disease and complex lesions, providing symptom relief, enhanced quality of life, and long-term survival benefits. In November 2023, the European Heart Journal published a subanalysis of the FRAGILE trial in Brazil, reporting that over 50% of frail patients improved to pre-frail status six months after coronary artery bypass graft surgery using heart-lung machines, with off-pump procedures uniquely enhancing grip strength and reducing unintentional weight loss. No significant differences were observed between on-pump and off-pump groups for other frailty parameters.
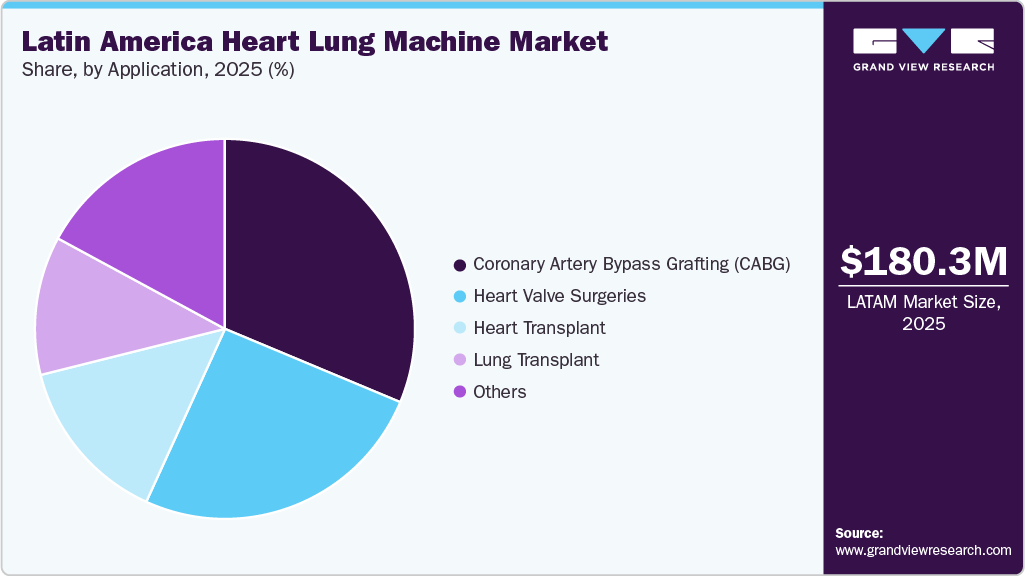
The heart valve surgeries segment is expected to grow at the fastest CAGR over the forecast period. In Latin America, heart valve surgeries are performed in specialized cardiac centers with advanced heart-lung machines, membrane oxygenators, and comprehensive monitoring systems. The expansion of healthcare infrastructure and the increasing prevalence of valvular heart disease drive demand for these procedures. Mechanical and biological valves are used, with selection based on patient age, lifestyle, and long-term anticoagulation needs. In April 2024, an editorial in the Brazilian Journal of Cardiovascular Surgery highlighted Brazil’s legacy in cardiac surgery and the development of cardiopulmonary bypass technology, including heart-lung machines. The article emphasized São Paulo’s role as a training hub for Latin America, with referral centers performing tens of thousands of procedures annually and contributing to innovations in surgical techniques, education, and patient care.
Country Insights
Brazil Heart Lung Machine Market Trends
Brazil dominated the market with a share of 34.68% in 2024. Brazil’s demand is increasing as the country expands its cardiovascular care capacity and enhances access to advanced cardiac surgery within both public (SUS) and private hospital systems. The growing incidence of coronary artery disease, more referrals to tertiary cardiac centers, and growing use of minimally invasive and hybrid procedures are pushing hospitals to modernize perfusion equipment. In September 2025, Mayo Clinic Proceedings reported on iCardio, a Brazilian real-world data platform that analyzes over 375,000 cardiovascular procedures, including heart surgeries and interventional cardiology, across 558 healthcare centers.
Mexico Heart Lung Machine Market Trends
Mexico market is expected to grow over the forecast period. Mexico’s market is expanding as cardiac care reaches more states and hospitals increase capacity for bypass surgery and advanced perfusion support. The rise in lifestyle-driven heart disease, earlier referrals, and growing surgical volumes are pushing facilities to upgrade technology. Growth in private hospital networks and medical tourism near established cities and tourism hubs is also shaping demand, as providers seek systems that align with international care expectations.
Key Latin America Heart Lung Machine Companies Insights
Key players operating in the Latin America heart lung machine market are undertaking various initiatives to strengthen their presence and increase the reach of their products and services. Strategies such as expansion activities and partnerships are key in propelling the market growth.
Key Latin America Heart Lung Machine Companies:
Medtronic
Terumo Corporation
LivaNova PLC
Getinge AB
Braille Biomedica
Nipro Corporation do Brasil (NIPRO)
Surge Cardiovascular
EUROSETS
Technowood America Corporation
Fresenius Medical Care
MERA (Senko Medical Instrument Mfg. Co., Ltd.)
Recent Developments
In January 2025, HIC in Colombia performed off-pump coronary bypass surgery for an international patient, underscoring rising regional expertise in techniques that avoid heart-lung machines and reinforcing Colombia’s position as a competitive destination for advanced cardiovascular surgery and medical tourism.
In October 2024, Medtronic received U.S. FDA clearance for the Affinity Fusion oxygenator, designed for use in cardiopulmonary bypass procedures. The device features an integrated arterial filter to help reduce micro-emboli and enhance patient safety during CPB. It incorporates balance biosurface technology to improve biocompatibility and minimize blood activation when in contact with artificial surfaces. This clearance reinforces its clinical role as a next-generation, single-use oxygenator for open heart surgeries.
In May 2023, Getinge AB launched the Rotaflow II ECMO system in Brazil, introducing advanced extracorporeal life support technology with features not previously available in the country. The new, lighter, and more compact device aims to support critically ill patients with severe cardiac or respiratory failure while improving usability for hospital staff.
Latin America Heart Lung Machine Market Report Scope
Report Attribute
Details
Market size value in 2025
USD 104.23 million
Revenue forecast in 2033
USD 150.64 billion
Growth rate
CAGR of 4.71% from 2025 to 2033
Actual Data
2021 – 2024
Forecast period
2025 – 2033
Quantitative units
Revenue in USD million/billion and CAGR from 2025 to 2033
Report coverage
Revenue forecast, company ranking, competitive landscape, growth factors, and trends
Segments covered
Product, application, end use, country
Regional scope
Latin America
Country scope
Brazil; Mexico; Argentina; Colombia; Chile
Key companies profiled
Medtronic; Terumo Corporation; LivaNova PLC; Getinge AB; Braille Biomedica; Nipro Corporation do Brasil (NIPRO); Surge Cardiovascular; EUROSETS; Technowood America Corporation; Fresenius Medical Care; MERA (Senko Medical Instrument Mfg. Co., Ltd.)
Customization scope
Free report customization (equivalent up to 8 analysts working days) with purchase. Addition or alteration to country, regional & segment scope.
Pricing and purchase options
Avail customized purchase options to meet your exact research needs. Explore purchase options
Latin America Heart Lung Machine Market Report Segmentation
This report forecasts revenue growth at the regional and country levels and provides an analysis of industry trends in each sub-segment from 2021 to 2033. For this study, Grand View Research has segmented the Latin America heart lung machine market report based on product, application, end use, and country:
Product Outlook (Unit Volume in ‘000 Units; Revenue in USD Million, 2021 – 2033)
Capital Equipment
Consumables
Application Outlook (Unit Volume in ‘000 Units; Revenue in USD Million, 2021 – 2033)
End Use Outlook (Unit Volume in ‘000 Units; Revenue in USD Million, 2021 – 2033)
Country Outlook (Unit Volume in ‘000 Units; Revenue in USD Million, 2021 – 2033)
Brazil
Mexico
Argentina
Colombia
Chile


