Table of Contents
Trial design
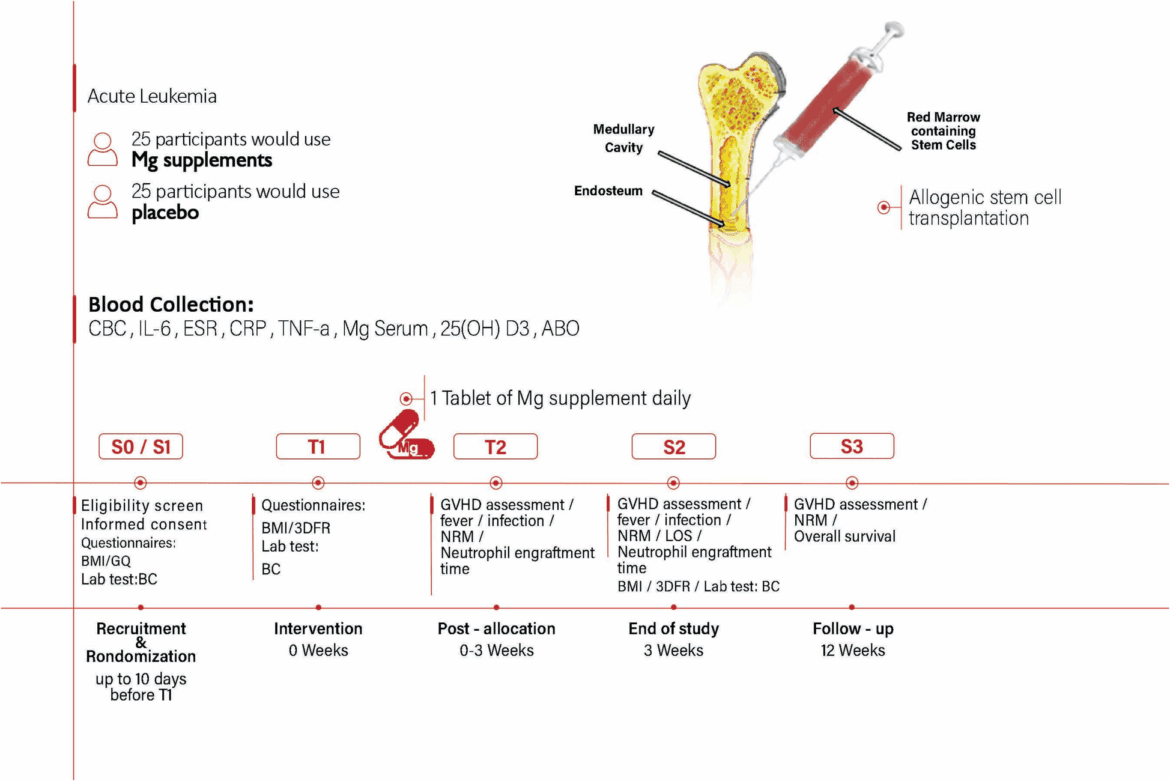
This study will be a controlled, randomized clinical trial with an intervention period of 3 weeks (Fig. 1). The trial was registered at the Iranian Registry of Clinical Trials at https://irct.behdasht.gov.ir (Identifier: IRCT20240812062726N1). In this article, we will use the Standard Protocol Items: Recommendations for Interventional Trials (SPIRIT) to present both the findings and specifics of the study protocol, such as the timing of participant enrollment, interventions, and assessments [15].
Overview of study timeline. Abbreviations: BMI body mass index, BC blood collection, CBC complete blood cell parameters, IL-6 Interleukin-6, CRP C-reactive protein, ESR erythrocyte sedimentation rate, TNF-α tumor necrosis factor-alpha, 25 (OH) D3 25-hydroxy vitamin D3, ABO ABO blood group system, GQ general questionnaire, 3DFR three-Day Food Recall, NRM non-relapse mortality, GVHD graft-versus-host disease, LOS length of stay
Trial population
Participants aged 18 to 60 years with acute leukemia admitted to Talaghani Hospital’s Bone Marrow Transplantation department will be eligible for inclusion. The subjects must satisfy all inclusion criteria and no exclusion criteria presented in Table 1 to be eligible for the trial. The study is a randomized controlled trial, and recruitment will begin in May 2025 and is expected to be completed approximately by the end of May 2026.
Sample size calculation
The sample size was determined using G*Power software (version 21.0.3) based on anticipated differences in TNF-α levels between groups. Using two-sample t-tests with reference to Lv et al. [16], who reported a mean difference of 15.38 pg/mL and pooled standard deviation of 4.37 pg/mL in TNF-α levels, and with α = 0.05 (two-tailed), power = 0.80, and allocation ratio of 1:1, the minimum required sample size was calculated as 21 patients per group. Considering a potential dropout rate of 10%, the study aims to recruit 25 patients in each group (total sample size = 50 patients.
Randomization
Randomization in this study will be performed using the Permuted Block Randomization method through Statistical Package for Social Sciences (SPSS) random number generator software. Prior to randomization, participants will be stratified by age groups (18 to 40 and 40 to 60 years) and gender (male/female). Block sizes will vary randomly between 4, 6, and 8 participants and will remain concealed from the recruitment team to maintain randomization integrity. A separate research methodologist will oversee the sequence generation process. The allocation will be conducted using variable block sizes (4, 6, 8) within each stratum to ensure balance between groups, with possible sequence patterns. The block size pattern will remain concealed from the recruitment team to maintain randomization integrity.
Participant enrollment will be conducted by trained research team members who will remain blinded to the allocation sequence. The allocation process will be managed independently to prevent selection bias, with codes stored securely and accessed only when each new participant is enrolled.
Intervention
The intervention will be initiated following stratified randomization. During hospitalization in the bone marrow transplantation unit, the intervention group will receive 420 mg of magnesium citrate capsules once daily after breakfast, while the control group will receive 420 mg of inert compounds containing starch capsules once daily after breakfast for 3 weeks. This timing with food was chosen to optimize magnesium absorption and minimize potential gastrointestinal side effects. Both supplements will be delivered by the ward nurses according to hospital medication protocols. Concomitant magnesium supplementation is prohibited during the intervention period.
The supplements will be provided in identical packages to maintain blinding. All participants will be monitored daily by the medical team for potential adverse events and laboratory findings during the intervention period. Any abnormal findings will be addressed according to standard hospital protocols.
The study timetable includes the following steps (Table 2):
S0 & S1: “Recruitment” and “ Randomization“—The assessment of inclusion and exclusion criteria, completion of study questionnaires, obtaining informed consent, completing the demographic questionnaire, Body Mass Index (BMI), laboratory tests (CBC, ESR, CRP, IL-6, TNF-α, Mg serum, 25(OH)Vit D3, ABO), determining the stem cell donor, and chemotherapy diet will be performed at admission to the transplant unit.
T1: Baseline assessment (Day 0)—The three-day Food Recall (3DFR) will be completed.
T2: Post-allocation (Days 0–21)—During hospitalization in the transplant unit, daily evaluation of occurrence and grade of GVHD, fever, infection, engraftment time, non-relapse mortality (NRM), and serum Mg levels will be performed. The magnesium/placebo supplement will be delivered by ward nurses once daily after breakfast.
S2: End of study (Day 21)—BMI, 3DFR, and laboratory tests will be reassessed before discharge.
S3: Follow-up (week 12)—At first post-discharge clinic visit, participants will be evaluated for GVHD, fever, infection occurrence, and will be informed of their test results.
Blinding
The blinding process will be managed by an independent third-party pharmacist responsible for the packaging and labeling of study supplements. Each package will be assigned a unique identification number, stored securely and separately from randomization codes. The magnesium supplement and placebo will be identical in color, smell, and packaging to maintain appropriate blinding. Neither the participants, investigators, research staff, nor outcome assessors will have access to treatment assignments until study completion. Randomization codes will be stored in sealed envelopes in a locked cabinet accessible only to the principal investigator. Emergency code breaking will be documented with participant ID, date, reason, and responsible person. Emergency unblinding will only be permitted for serious adverse events requiring immediate medical intervention, with principal investigator approval and complete documentation of the reason and timing.
Adherence to the intervention
Adherence to the intervention will be checked by face-to-face assessments during hospitalization in the bone marrow transplantation unit. The supplement consumption will be delivered by the ward nurses according to hospital medication protocols, and compliance will be documented in the patient’s registration file. A compliance rate of less than 80% for the supplement consumption will be considered as low adherence.
Recruitment and consent
Eligible patients will be identified through daily screening of acute leukemia admissions to the bone marrow transplantation ward at Taleghani Hospital. The research team will approach potential participants during pre-transplant consultations and provide detailed study information. Written informed consent will be obtained after allowing adequate time for consideration and questions. Recruitment strategies include collaboration with the hematology team for early identification of candidates and flexible scheduling to accommodate patient preferences. Based on historical admission data of approximately 4–5 eligible patients per month, recruitment is expected to be completed within the planned 12-month timeframe.
Data monitoring
The trial will be coordinated by the Nutrition Research Center at Shahid Beheshti University of Medical Sciences. The Trial Management Group includes the principal investigator, co-investigators, research team members, and statistician. This group will monitor all aspects of trial conduct and progress, adherence to the trial protocol, and participant safety. Given the low-risk nature of the intervention and short study duration, no independent data monitoring committee or steering committee was deemed necessary.
Manufacture of study supplements
The magnesium with a chemical formula of C6H6MgO7, purity ≥ 99%, and low metal ion content will be procured from Ofogh Tolid Darou Pars Co., Ltd.
Adverse event reporting and management
This trial is considered low-risk as magnesium citrate supplementation presents minimal risk to participants. Mild to moderate gastrointestinal disturbances such as diarrhea, nausea, and abdominal bloating may occur. To minimize these effects, nutritional and clinical conditions will be evaluated regularly. Serious Adverse Events (SAEs) will be reported to the principal investigator within 24 h and include death, life-threatening conditions, or unexpected hospitalizations. The sponsor will notify the Research Ethics Committee of any serious breaches of Good Clinical Practice principles. If any gastrointestinal complications occur, the dose of magnesium supplement will be reduced to 210 mg daily, or temporary discontinuation will be considered based on the severity of symptoms. Criteria for intervention discontinuation include: serious adverse events, withdrawal of consent, disease relapse, ICU admission, or serum magnesium levels outside the 1.5–2.4 mg/dL range. All modifications will be documented and analyzed per the intention-to-treat principle [17].
Outcome measures
The primary efficacy outcome will be serum levels of TNF-α as the main inflammatory factors, measured at baseline (admission to the bone marrow transplantation unit) and after the intervention period (end of week 3).
The secondary outcomes will include:
1.
Serum IL-6, CRP and ESR levels, evaluated at baseline and after 21 days of transplantation
2.
The occurrence and grade of GVHD, which will be monitored daily during hospitalization continued through day 100 post-HSCT
3.
Fever occurrence, which will be recorded daily during hospitalization through temperature measurements
4.
Post-transplant infection, which will be evaluated during daily during hospitalization through day 100 post-HSCT
5.
NRM, which will be monitored from admission through day 100 post-HSCT, defined as death from any cause other than disease relapse
All blood samples will be analyzed in the laboratory unit of the department, and all adverse events will be recorded in detail in the patient’s files.
Measurements
Baseline assessment: After signing the written informed consent, a general questionnaire (GQ) will be completed using patient interviews and medical records. This questionnaire includes gender, age, education level, smoking and alcohol consumption, type of leukemia, stem cell donor source, number of donated CD34 + and CD3 + cells, underlying diseases, type of GVHD preventive drug treatment, and type of conditioning chemotherapy regimen.
Anthropometric measurements: Weight will be measured in a fasting state, without shoes, with minimal clothing using a digital scale (Seca, Hamburg, Germany) with 100 g accuracy. Height will be measured in a standing position without shoes, with heels touching the wall, using a wall-mounted tape with 0.5 cm accuracy. BMI will be calculated by dividing weight (kg) by height squared (m2) [18].
Laboratory factors: Blood samples (10 mL) will be obtained and serum will be isolated by centrifugation for 10 min at 3500 rpm. The serum will be maintained at − 80 °C until analysis. The following parameters will be measured [18]:
1.
Inflammatory factors (IL-6, TNF-α, CRP) using ELISA kits [19, 20]
2.
ESR using Westergren method [21]
3.
Complete Blood Count (CBC)
4.
Serum magnesium levels
5.
25-hydroxyvitamin D3 levels
6.
ABO blood type
Dietary assessment: 3DFR (two non-consecutive weekdays and one weekend day) will be completed at baseline. Food portions will be converted to grams using the household measures guide. Dietary data will be analyzed using Nutritionist-IV (N4) software program (version 7.0; N-Squared Computing, Salem, OR, USA) to evaluate:
1.
Total energy intake
2.
Macronutrients (carbohydrates, proteins, fats)
3.
Micronutrients (magnesium, selenium, vitamins D, E, and C)
Clinical monitoring: Engraftment time will be assessed through neutrophil engraftment, defined as the first of three consecutive days with absolute neutrophil count (ANC) > 0.5 × 109 [22]. During hospitalization, patients will be monitored for infection and fever, and the length of hospital stay after HSCT will be recorded. After discharge, patients will be followed monthly until 100 days after HSCT for GVHD and its type and grade, as well as survival and NRM. GVHD will be evaluated based on Glucksberg criteria, assessing the main affected organs, including skin, gastrointestinal tract, liver, and lymphoid tissue (Table 1 supplementary) [23]. All these parameters will be monitored through medical records and regular follow-up at the transplant center.
Data management
In this study, study questionnaires (general questionnaire, 3-day food recall, and anthropometric measurements) in Word format were completed by trained researchers interviewing the patients in person during hospitalization. The subjects had the opportunity to review their responses for accuracy. The samples collected were blood samples taken by qualified phlebotomists. The principal investigator confirmed data entry while other contributing researchers checked the data for accuracy and integrity. The gathered data were organized into two datasets: (1) questionnaires that were filled in Microsoft Word and converted to PDF; (2) blood laboratory findings and inflammatory marker results provided by laboratories in PDF format. The data were summarized in a Microsoft Excel sheet. All files were stored under unique identification codes of the study participants. For privacy and confidentiality reasons, the only way to identify the patients’ data was to search the specific identification code assigned to each patient in the informed consent. Internal trial monitoring will be conducted by the institutional research office to ensure protocol compliance and data quality. Also, protocol modifications will be submitted to the Ethics Committee for approval and updated in the trial registry. Participants will be informed of relevant changes.
Statistical analysis
SPSS-24 software will be used for data analysis with a significance level set at p < 0.05. Data normality will be evaluated using the Kolmogorov-Smirnov test. Quantitative data will be presented as mean ± standard deviation or median (interquartile range), and qualitative data as frequency (percentage). Chi-square test or Fisher’s exact test will be used to compare qualitative variables between groups. For quantitative variables, independent t-test will be used for normal distributions and Mann-Whitney test for non-normal distributions. Regression analysis will be used to investigate the relationship between serum biomarkers and anthropometric indicators while adjusting for confounding factors. The intention-to-treat principle will form the basis of the statistical analysis. Missing data will be handled using complete case analysis for primary outcomes. If missing data exceeds 5%, sensitivity analysis using multiple imputation will be performed to assess the robustness of results.